Sponsor: National Institute of Nursing Research
07/01/2012 - 06/30/2014
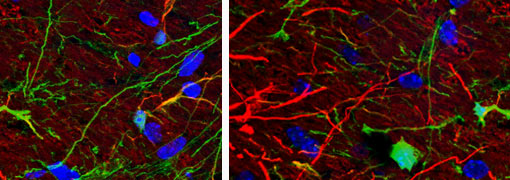
Existing data support a critical role of neurokinin 1 receptor (NK1 receptor) expressing neurons in the  dorsal spinal cord in mediating pain processing. Taking the advantage of newly developed nanoparticles, we plan to explore whether conjugation of nanoparticles with a specific ligand to NK1 receptors, can achieve cell-type specific delivery of small interference RNA (siRNA) into NK1 receptor expressing cells upon ligand-induced endocytosis, and if so, whether this approach can lead to effective gene silencing and blocking behavioral hypersensitivity in vivo.
dorsal spinal cord in mediating pain processing. Taking the advantage of newly developed nanoparticles, we plan to explore whether conjugation of nanoparticles with a specific ligand to NK1 receptors, can achieve cell-type specific delivery of small interference RNA (siRNA) into NK1 receptor expressing cells upon ligand-induced endocytosis, and if so, whether this approach can lead to effective gene silencing and blocking behavioral hypersensitivity in vivo.
Sponsor: Roman Reed Spinal Cord Injury Research Fund of California
02/2012 - 02/2013
Chronic pain in spinal cord injury patients is difficult to manage, 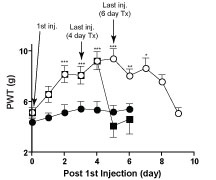 and affects the quality of the daily-lives adversely. Current medications can only provide partial pain relief and are often associated with intolerable side effects. The development of target specific, and safer pharmacological agents depends on the identification of new targets that mediate the development of chronic spinal cord injury pain.
and affects the quality of the daily-lives adversely. Current medications can only provide partial pain relief and are often associated with intolerable side effects. The development of target specific, and safer pharmacological agents depends on the identification of new targets that mediate the development of chronic spinal cord injury pain.
Dr. Luo’s team will study on whether interactions of two injury-induced proteins: thrompospondin-4 (TSP4) and the calcium channel alpha-2-delta-1 subunit proteins, play a critical role in spinal cord injury pain processing, and if so, whether it is mediated in a sensory-neuron-specific manner. Models with normal or null alpha-2-delta-1 gene expression, either in the spinal cord and dorsal root ganglia, or in subpopulations of sensory neurons, will be used to examine whether there is a correlation between levels of TSP4 expression and behavioral sensitivities to stimuli. Findings from these studies will provide us with critical information for the development of the next generation of analgesic agents for better chronic pain management.
| Back to Top |
Sponsor: National Institute of Neurological Disorders and Stroke
02/01/2010 - 01/31/2014
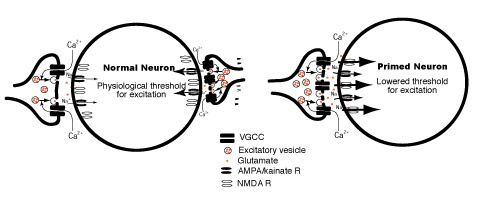
Existing data indicate that peripheral nerve injury induces an elevated level of the voltage-gated calcium channel CaVα2δ1 subunit proteins in dorsal root ganglia and spinal cord that contributes to neuropathic pain development through an unknown mechanism. We hypothesize that this injury-induced neuroplasticity may lead to abnormal synaptic transmission that plays a causal role in neuropathic pain processing. We are testing this hypothesis with multidisciplinary approaches. Completion of this study will provide important information for the understanding of neuropathic pain mechanisms and the development of next generation of neuropathic pain medications.
| Back to Top |
Sponsor: National Institute of Dental & Craniofacial Research
04/07/2011 - 02/29/2016
Chronic orofacial pain is a common clinical syndrome lacking specific and effective therapeutic agents due to the fact that cellular mechanisms of chronic orofacial pain are poorly understood. Based on data from gene chip microarray and chronic pain disorders derived from trigeminal nerve injuries, we hypothesize that trigeminal nerve injury induces thrombospondin-4 (TSP4) expression in trigeminal ganglia (TG) and associated brainstem/upper cervical spinal cord (Vc/C2) that causes sensory neuron hyperexcitability, and abnormal synaptogenesis in the trigeminal complex in the spinal cord. These changes underlie the transition from trigeminal nerve injury to chronic pain development. We plan to identify the critical domain(s) of TSP4 in mediating behavioral hypersensitivity and spinal neuron hyperexcitability, and the TSP4’s action site in chronic pain processing. Multidisciplinary approaches, including behavioral pharmacology, cellular and molecular biology, electrophysiology, confocal and electron microscopy, are used to determine the extent of abnormal synaptogenesis, signaling pathways in mediating the effects of TSP4 on sensory neuron excitability, spinal sensitization, and behavioral hypersensitivity. The final goal of these studies is to identify the peripheral and/or central mechanisms underlying TSP4-mediated transition to chronic pain states after trigeminal nerve injury.
| Back to Top |